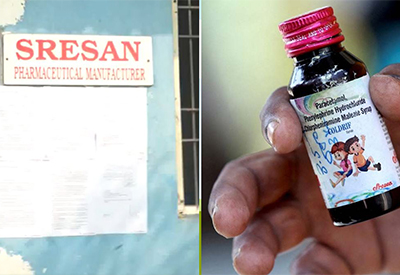
The Tamil Nadu health department has cancelled all manufacturing licenses of Sresan Pharma, following a tragic linkage between its cough syrup Coldrif and 24 child deaths.

Glimpse:
Authorities have ordered immediate closure of all Sresan Pharma units and license revocations across the board. The decisive move comes amid mounting public outcry and pressure on regulators to act.
In a stern regulatory response, Tamil Nadu’s government has formally revoked all manufacturing licenses of Sresan Pharmaceuticals and ordered the firm’s closure. The decision comes in the wake of reports linking Coldrif syrup to 24 child deaths across Madhya Pradesh.
Sresan’s plants are being shut down, and compliance teams will audit all related premises. Authorities are also working to trace and seize remaining stocks. The action is part of a wider clampdown manufacturers across implicated states are under scrutiny.
Regulators hope this bold response restores public trust and signals zero tolerance for lapses in pharmaceutical safety. Yet industry watchers caution that deeper systemic reform is needed to prevent recurrence.
“Revoking licenses is necessary, but rebuilding system integrity is the real task ahead.”
By
HB Team