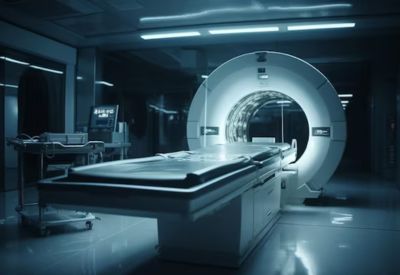
Philips has launched a trio of next-gen imaging innovations the AI-powered Verida spectral CT system, a helium-free 3.0 T MRI platform called BlueSeal Horizon MRI, and expanded availability of the radiation-free light-based navigation tool LumiGuide 3D Device Guidance aimed at delivering sharper diagnostics, faster workflows and safer, lower-radiation image-guided procedures.
Glimpse:
Philips’ new AI-enabled MRI and CT systems use dual-AI engines and deep-learning reconstruction to speed up imaging, improve clarity, and shorten scan times, allowing hospitals to handle more patients without compromising diagnostic accuracy. Alongside this, the LumiGuide platform leverages fiber-optic real-shape technology, which tracks light through a specialized guidewire to generate real-time, full-color 3D navigation images. This approach reduces radiation exposure by more than half and makes procedures significantly faster compared to traditional X-ray guidance. Together, these advancements signal a major shift toward safer, quicker, and more efficient radiology and interventional care, with the potential to transform hospital workflows globally.
Global health-tech firm Philips has announced an upgrade to its imaging and image-guided therapy portfolio unveiling three major innovations at once. The new offerings combine AI, sustainability (helium-free MRI), speed, and radiation-free navigation to address both diagnostic and procedural needs in modern healthcare.
Verida spectral CT system stands out as the world’s first detector-based spectral CT system powered end-to-end by AI. According to Philips, the system’s AI-enabled reconstruction generates up to 145 images per second and can complete a full study in under 30 seconds nearly double the throughput of many traditional CT machines. The high-speed capability makes it ideal for high-volume imaging centres. The system also claims substantial reductions in radiation exposure and energy consumption (up to 45%), making CT imaging both safer and greener.
BlueSeal Horizon 3.0 T MRI marks a shift away from traditional MRI dependencies on liquid helium. With a helium-free magnet, the system simplifies installation, avoids helium-related operational disruptions, and supports high-field MRI in settings where helium supply or infrastructure may be limiting. On top of that, Philips is integrating AI-driven workflows (accelerated scanning, automated planning, real-time scan previews, denoising/artifact reduction) to cut scan times, improve image clarity, and boost radiologist efficiency. This could greatly expand MRI access especially in hospitals that previously found high-field MRI difficult to deploy.
Perhaps the most transformative LumiGuide 3D Device Guidance expands commercial availability across Europe and the US, bringing radiation-free, light-based navigation to more hospitals. LumiGuide uses fiber-optic guidewires (powered by the proprietary FORS technology) to deliver full-color, real-time 3D visualization of devices inside the body effectively replacing continuous X-ray fluoroscopy for many image-guided procedures. Integrated with Philips’ Azurion platform, LumiGuide promises enhanced procedural precision, reduced radiation exposure (to near-zero fluoroscopy usage in many cases), faster procedure times and improved safety for patients and clinical staff. Early users report up to 37% faster procedures and as much as 56% reduction in dose-area product (DAP) compared to conventional X-ray navigation.
Together, these innovations reflect a broader strategic push from Philips combining AI, sustainable design (helium-free magnets), and next-gen navigation aimed at redefining diagnostic imaging and interventional radiology workflows. The company hopes these advances will help hospitals deliver faster, safer, more efficient care while reducing operational constraints such as helium supply, radiation exposure, workflow bottlenecks, and energy consumption.
Time Medical International Ventures (India) Private Limited a wholly owned subsidiary of Fischer Medical has entered into a global supply agreement with Indonesian partner PT Pharmindo Rimpang Kokoh (PT Pariko) to deliver 250 AI-enabled portable digital X-ray systems. The total contract value is approximately US$ 6.5 million. nits are being supplied as part of a public-health initiative in Indonesia aimed at supporting the country’s tuberculosis (TB) eradication and lung-disease surveillance programmes. The deployment targets remote, underserved and rural areas where access to fixed radiology infrastructure is limited enabling mobile or campaign-based screening for TB and other pulmonary illnesses.
The rollout is planned in phased tranches, with the full delivery slated to complete by March 2026. Besides delivering hardware, Fischer Medical’s subsidiary will provide technical support and ensure integration of AI-supported diagnostics which helps radiologists identify lung abnormalities, possibly improving the speed and accuracy of diagnosis in large population-level screening.
For Fischer Medical, this international order strengthens its position as a key med-tech supplier beyond India, demonstrating capability in manufacturing and supplying advanced imaging solutions for global public-health challenges. The deal aligns with the company’s broader strategy: after obtaining regulatory license for MRI manufacturing in India earlier this year, it is now expanding its footprint across Southeast Asia via sale of portable imaging systems.
“By combining AI-powered imaging, helium-free MRI, and light-based navigation, we’re not just evolving technology we are transforming how care is delivered making it faster, safer and more sustainable.”
By
HB Team