The World Health Organization has issued a global health advisory after tests found three Indian cough syrups tainted with dangerous levels of diethylene glycol (DEG), linked to child fatalities.
Glimpse:
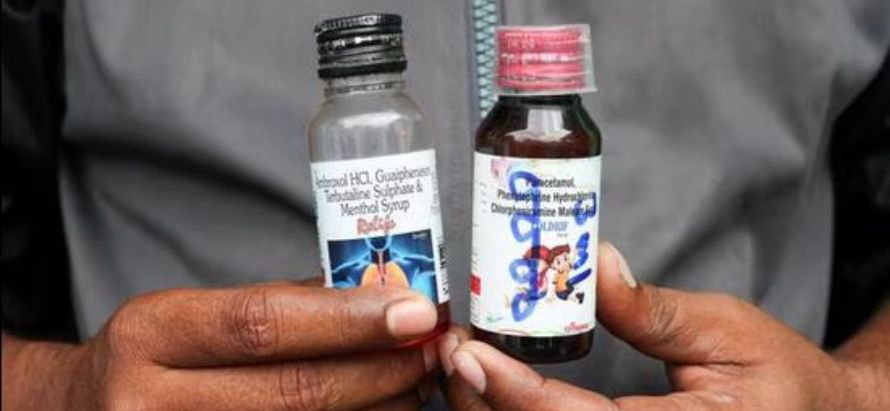
The syrups Coldrif, Respifresh TR, and ReLife have been declared contaminated and recalled. Although none were officially exported, WHO warns of risks from informal distribution. Regulators worldwide are urged to monitor entry points and report any detections.
In a sharp escalation of the ongoing pharmaceutical safety crisis, the World Health Organization (WHO) has issued a global alert concerning three Indian cough syrups found to be severely contaminated. The products Coldrif, Respifresh TR, and ReLife were detected in lab tests to contain diethylene glycol (DEG) at levels orders of magnitude above safe thresholds.
While India’s CDSCO confirmed these syrups were not exported through formal channels, the WHO cautioned that informal trade routes could facilitate their spread across borders. National drug regulatory agencies are being urged to launch targeted surveillance, conduct random sampling of oral liquid medicines from the implicated manufacturing sites, and report any findings promptly.
Health authorities in India have already initiated recalls, halted production, and frozen batches in circulation. States have been instructed to intensify drug inspections, particularly in rural and informal supply chains. The alert underscores gaps in regulatory enforcement and points to the need for deeper reforms in pharmaceutical safety systems.
“Safety in medicine cannot stop at borders a tainted drug anywhere is a threat everywhere.”
By
HB Team























