Singapore-based healthtech startup Aevice Health has secured regulatory approval in Australia for its AI-powered wearable respiratory monitoring system for children aged three and above marking a milestone for continuous pediatric respiratory care outside the clinic.
Glimpse:
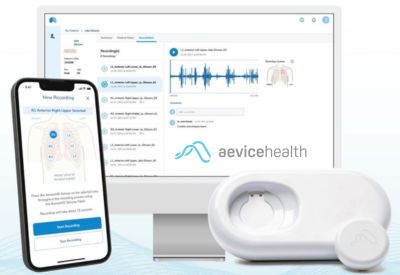
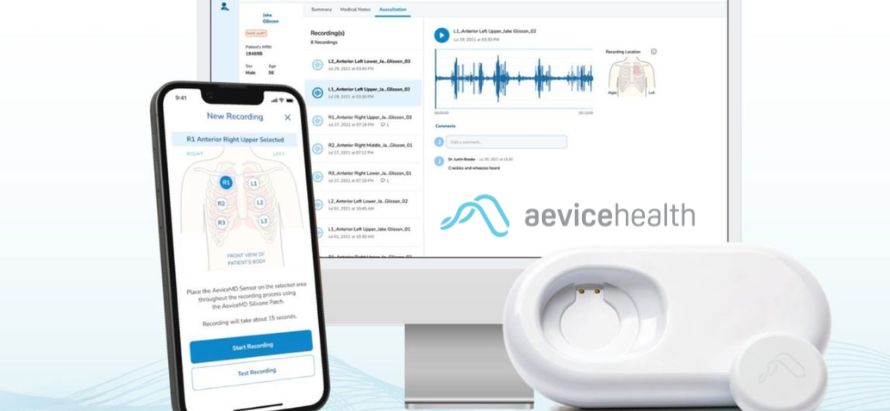
Approved by Australia’s Therapeutic Goods Administration, AeviceMD integrates digital auscultation, wheeze detection, and heart and respiratory rate measurement into a single Class IIa device. Backed by clinical evidence and prior U.S. clearance, the system aims to enable early detection and remote management of pediatric respiratory conditions such as asthma.
Singapore-based Aevice Health has received approval from Australia’s Therapeutic Goods Administration (TGA) for its AI-powered respiratory monitoring system, extending use to individuals aged three years and older. The clearance positions AeviceMD as the first wearable stethoscope approved in Australia for pediatric use, supporting continuous respiratory monitoring beyond traditional clinical settings.
AeviceMD consolidates multiple functions digital auscultation, wheeze detection, and heart and respiratory rate measurement into a single Class IIa medical device. Designed with pediatric care in mind, the wearable supports children with fluctuating respiratory conditions such as asthma, where symptoms can worsen between clinic visits and early intervention is critical.
The approval was supported by data from an observational study conducted with National University Hospital, where the device demonstrated improved performance and accuracy in detecting wheezing among children. Pilot deployments further indicated strong potential for continuous monitoring and early identification of symptom deterioration, helping clinicians act sooner.
AeviceMD pairs with mobile and web applications that allow clinicians to monitor patients remotely, reducing the need for frequent in-person visits while enabling timely intervention when symptoms change. This model aligns with outpatient and remote patient monitoring pathways, particularly for chronic pediatric respiratory care.
Australia’s approval follows Aevice Health’s earlier regulatory clearance from the U.S. Food and Drug Administration for pediatric use of the same wearable stethoscope underscoring growing international confidence in the technology’s safety and effectiveness.
With the new TGA clearance, Aevice Health plans to explore pilot deployments and workflow integrations with local hospitals and respiratory specialists, focusing on embedding continuous respiratory monitoring into existing asthma management programs across outpatient care.
“With TGA clearance, we will work closely with hospitals and respiratory specialists to integrate continuous respiratory monitoring into outpatient asthma pathways supporting earlier intervention and better outcomes for children”
By
HB Team