Following lab alerts, Telangana’s Drugs Control Administration has issued a stop use order for two cough syrups found to be adulterated, intensifying the state’s response to the national medicine safety crisis.
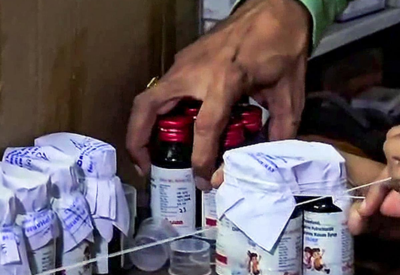
Glimpse:
Relife and Respifresh TR cough syrups have been flagged by the Telangana DCA as containing toxic contaminants. All existing stocks are to be frozen and removed from the supply chain. Distributors, retailers, hospitals, and pharmacies have been instructed to halt use immediately, as enforcement actions continue across the state.
In a swift regulatory move, the Telangana Drugs Control Administration has issued a statewide notice banning two cough syrups Relife and Respifresh TR after recent laboratory results detected harmful substances. The directive applies to all medical outlets: retailers, wholesalers, hospitals, and pharmacies must freeze stocks of the flagged batches and stop dispensing them.
The action comes in the wake of child fatalities in Madhya Pradesh and Rajasthan, where contaminated cough syrups (especially “Coldrif”) have been implicated. Telangana is treating this as a critical public health threat, mobilizing its drug inspectors to audit supplies, identify violations, and ensure compliance.
Officials emphasize that the ban is preventive: no new sales or use should happen unless further testing clears the batches. Underlying investigations are ongoing to trace the origin of contamination and hold responsible parties accountable.
“Medicine must heal not hurt. When contamination risks arise, decisive removal is the only safe path.”
By
HB Team